◆ Biological Evaluation
● Team Introduction

● Evaluation Services
In accordance with the guidelines of GB/T 16886, ISO 10993, OECD, and national or internationally recognized standards, we conduct biological evaluation of medical devices, microbiological testing, large and small animal surgical trials, veterinary pathology and clinical laboratory testing, and analytical chemistry services, providing safety and efficacy evidence for the preclinical research of medical devices.
Medical Device Biological Evaluation | Test Type | Test Item | Standard or Guideline |
In Vitro Cytotoxicity Test | Neutral Red Uptake (NRU) Cytotoxicity Test | GB/T 16886.5/ISO 10993-5 | |
Colony Forming Cytotoxicity Test | |||
MTT Cytotoxicity Test | |||
XTT Cytotoxicity Test | |||
Irritation and Sensitization Tests | Skin Irritation | GB/T 1688.23/ ISO10993-23/ OECD404/OECD405 | |
Intracutaneous Irritation | |||
Eye Irritation | |||
Oral Mucosal Irritation | |||
Rectal Irritation | |||
Penile Irritation | |||
Vaginal Irritation | |||
Guinea Pig Maximization Test (GPMT) | GB/T 16886.10/ ISO 10993-10/ OECD406 | ||
Buehler Closed Patch Test | |||
Skin Sensitization Test |
● Core Certifications/Accreditations/Qualifications
Lewwin Pharm Medical Device Examination and Testing Platform has obtained international OECD GLP Certification, US FDA GLP Certification, AAALAC International Full Accreditation, CNAS and CNCA GLP Certifications (China National Accreditation Service for Conformity Assessment, National Certification and Accreditation Administration), and national CMA Accreditation (China Metrology Accreditation). Currently, multiple device projects have been approved for marketing through US FDA review. We can support global regulatory submissions for Class II and Class III medical devices, innovative medical device products, and drug-device combination products.

Project Experience
Lewwin Pharm Medical Device Evaluation Products: Absorbable dura mater closure medical adhesive, absorbable vascular closure medical adhesive, α-cyanoacrylate medical adhesive, single use cannula trocars, and various drug-device combination products have all passed review by the National Medical Products Administration. Among them, those submitted for FDA registration have all been approved for marketing.
一次性套管针穿刺生物学评价
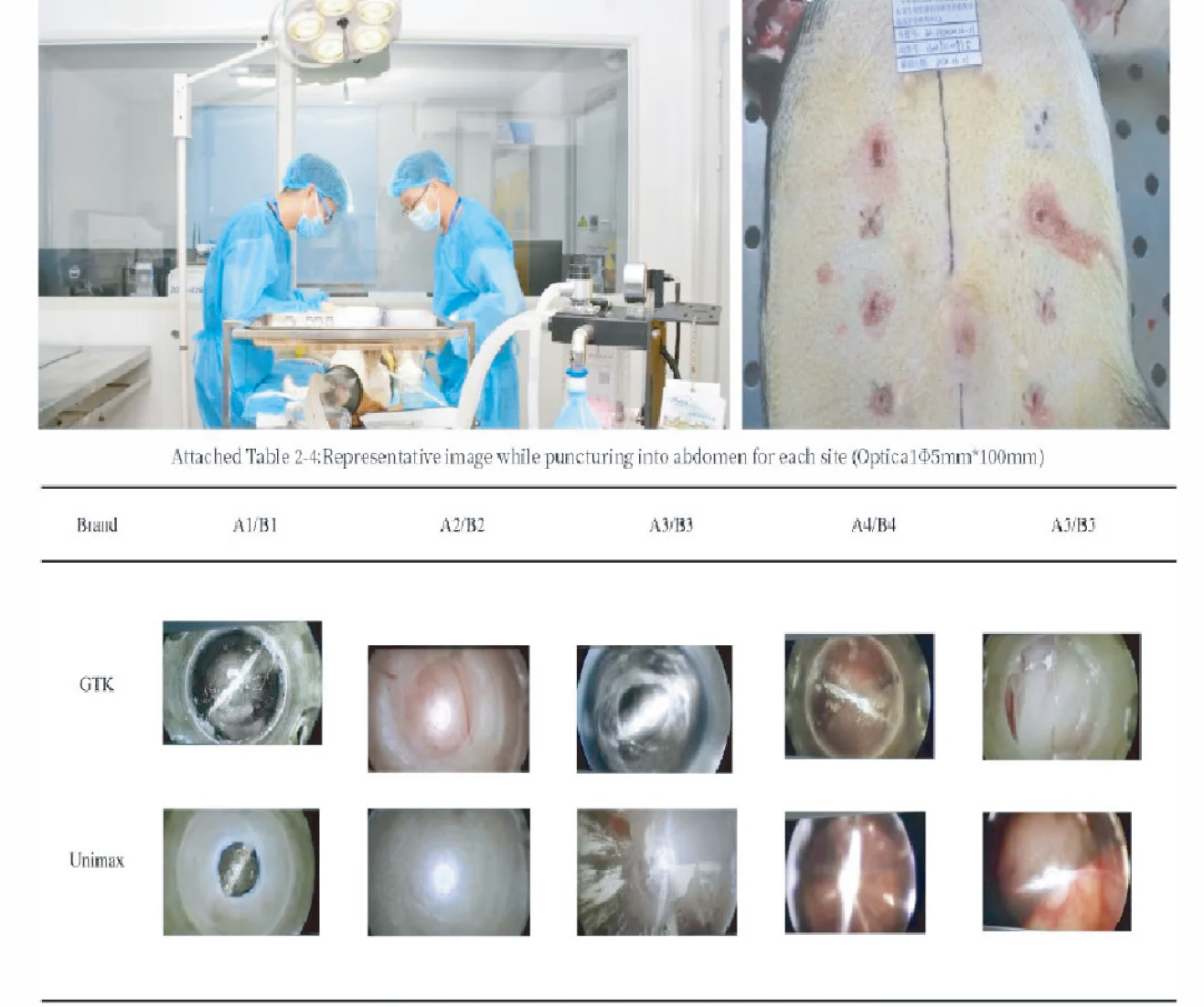
Imaging Rendering of Single Use Cannula Trocar
Medical Device Biological Evaluation | Test Type | Test Item | Standard or Guideline |
Systemic Toxicity Study | Acute Systemic Toxicity | GB/T 16886.11/ISO 10993-11 | |
Subacute Systemic Toxicity | |||
Subchronic Systemic Toxicity | |||
Chronic Systemic Toxicity | |||
Acute Oral Toxicity | OECD401/420/423/425 | ||
Acute Dermal Toxicity | OECD402 | ||
Acute Inhalation Toxicity | OECD403/436 | ||
28-Day Repeated Dose Oral Toxicity in Rodents | OECD407 | ||
90-Day Repeated Dose Oral Toxicity in Rodents | OECD408 | ||
90-Day Repeated Dose Oral Toxicity in Non-rodents | OECD409 | ||
21-Day/28-Day Repeated Dose Dermal Toxicity | OECD410 | ||
90-Day Subchronic Dermal Toxicity | OECD411 | ||
28-Day Subacute Inhalation Toxicity | OECD412 | ||
90-Day Subchronic Inhalation Toxicity | OECD413 | ||
Chronic Toxicity Study | OECD452 | ||
Reproductive Toxicity Study | Embryo-fetal Developmental Toxicity | GB/T 16886.3/ISO 10993-3/OECD414 | |
One-generation Reproductive Toxicity | GB/T 16886.3/ISO 10993-3/OECD415 | ||
Two-generation Reproductive Toxicity | GB/T 16886.3/ISO 10993-3/OECD416 | ||
Genotoxicity Test | Micronucleus Test | GB/T 16886.3/ISO 10993-3/OECD474/487 | |
Chromosomal Aberration | GB/T 16886.3/ISO 10993-3/OECD473/475 | ||
Bacterial Reverse Mutation (Ames Test) | GB/T 16886.3/ISO 10993-3/OECD471 | ||
Mouse Lymphoma Assay | GB/T 16886.3/ISO 10993-3/OECD490 | ||
Blood Interaction Test | Hemolysis | GB/T 16886.4/ISO 10993-4 | |
In Vivo Thrombosis Test | |||
Partial Thromboplastin Time (PTT) Test | |||
Complement Activation | |||
Implantation Test | Subcutaneous Implantation | GB/T 16886.6/ISO 10993-6 | |
Intramuscular Implantation | |||
Bone Implantation | |||
Brain Implantation | |||
Immunotoxicity Test | In Vitro T-lymphocyte Transformation Test | GB/T 16886.20/ISO/TS 10993-20 | |
Serum Immunoglobulin and Complement Component Assay (ELISA) | |||
Plaque-forming Cell Assay (Agar Solid-phase Method) | |||
Mouse Peritoneal Macrophage Phagocytosis of Chicken Red Blood Cells (Semi-in vivo Method) | |||
Determination of α-Gal Antigen Clearance Rate in Animal-derived Medical Devices | |||
Flow Cytometric Analysis of Splenic Lymphocyte Subsets in Animals | |||
● Evaluation Services
Test Items | |
Medical Device Microbiological Testing Items | Sterility Test |
Microbial Limit Test | |
Bacterial Endotoxin Test | |
Medical Device Large and Small Animal Surgical Trials | Provide medical device efficacy and safety reports compliant with NMPA GLP, FDA GLP, OECD GLP, CNAS CNCA GLP, and CMA requirements. |
World-class AAALAC-accredited animal housing facilities that meet the requirements for surgical and other animal experiments involving multiple species such as pigs, dogs, monkeys, rabbits, mice, and rats. | |
The operating rooms are equipped with professional devices including lift-type constant-temperature animal surgical tables, surgical shadowless lamps, MATRX large-animal ventilator-anesthesia machines, surgical microscopes, electrocautery knives, dental surgical equipment, brain stereotaxic instruments, cranial drills, ECG monitors, animal-dedicated ultrasound systems, 16-channel physiological recorders, Doppler hemodynamic systems, and EEG telemetry systems. These facilities provide a comprehensive supporting platform for conducting various evaluation services—such as general surgery, ophthalmology, neurosurgery, soft-tissue surgery, cardiac surgery, neurology, and dentistry—for both large- and small-animal medical devices. | |
Medical Device Analytical Chemistry Testing Services | Medical Device Chemical Characterization Leachable Qualitative and Quantitative Analysis Extractable Qualitative and Quantitative Analysis |
Medical Device-Drug Compatibility Study Leachable Study Extractable Study Method Development and Validation for Drug-Device Combination Products | |
Medical Device In Vitro Degradation Study Molecular Weight and Molecular Weight Distribution Method Development and Validation for Known Degradation Products Qualitative and Quantitative Analysis of Unknown Degradation Products | |
Veterinary Pathology and Veterinary Clinical Laboratory Testing Services for Medical Devices | Provide professional soft tissue and hard tissue processing, pathological diagnosis, and morphometric analysis services. |
Provide nearly 100 veterinary clinical laboratory test items (including: liver function, kidney function, myocardial enzymes, blood lipids, coagulation function, bone marrow cytomorphology, blood/body fluid testing, and routine urinalysis, etc.). | |
The team’s experience covers clinical testing, pathological diagnosis, and report writing for pharmaceutical and medical device products. The veterinary clinical testing and pathology technical team supports global clients in passing NMPA, FDA, and OECD reviews and registration applications. |


● Certificate
Organization | Verification Content | Result |
National Institutes for Food and Drug Control | Proficiency Testing for Pathological Evaluation of Medical Device Implantation Tests | Satisfactory-Excellent |


